International team finds high-resolution structural analysis of protein behind Huntington’s
From the WashU Newsroom…
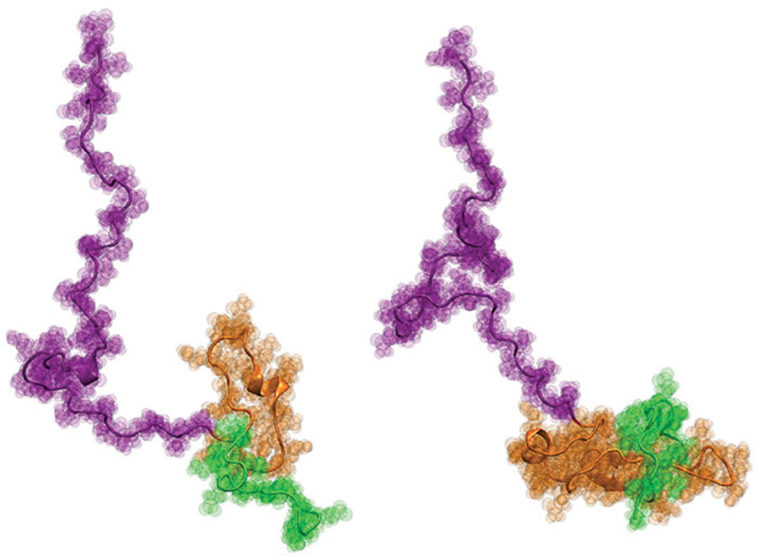
Huntington’s disease is a progressive, fatal neurodegenerative disorder that is caused by mutations in one specific gene called huntingtin (Htt). In the 20-plus years since the Htt gene was identified, researchers have focused on the protein encoded by the Htt gene, called Httex1. This protein accumulates in the brains of Huntington’s disease patients, and the prevailing hypothesis has been that it undergoes a dramatic structural change when a repetitive tract of the amino acid glutamine mutates into an aberrantly long region known as the mutationally expanded polyglutamine (polyQ) tract.
Now, for the first time, the team of Hilal A. Lashuel at Ècole Polytechnique Fèdèrale de Lausanne (EPFL) in Switzerland; Edward A. Lemke at the European Molecular Biology Laboratory (EMBL) in Germany; and Rohit V. Pappu at Washington University in St. Louis has uncovered a detailed structural description of Htt as a function of polyQ length. The work was published recently in the Journal of the American Chemical Society.