Years before symptoms of Alzheimer’s disease appear, two kinds of damaging proteins silently collect in the brain: amyloid beta and tau. Clumps of amyloid accumulate first, but tau is particularly noxious. Wherever tangles of the tau protein appear, brain tissue dies, triggering the confusion and memory loss that are hallmarks of Alzheimer’s.
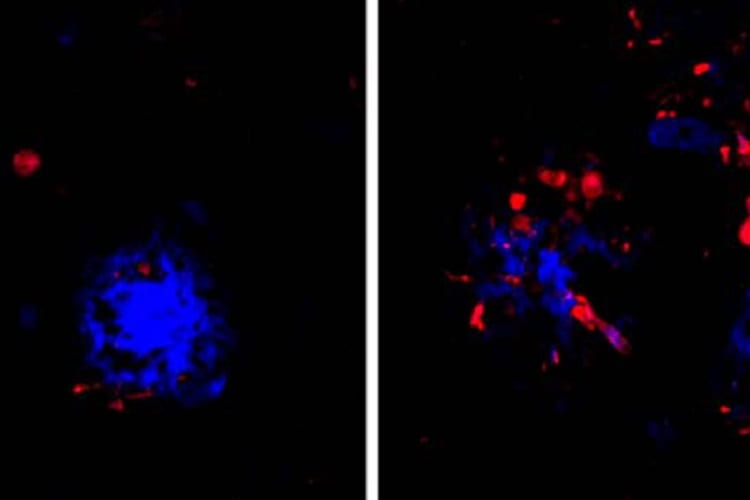
Now, researchers at Washington University School of Medicine in St. Louis have found that the link between the two proteins may lie in the brain’s immune cells that hem in clumps of amyloid. If the immune cells falter, amyloid clumps, or plaques, injure nearby neurons and create a toxic environment that accelerates the formation and spread of tau tangles, they report.
The findings, in mice and in people, are published June 24 in Nature Neuroscience. They suggest that reinforcing the activity of such immune cells – known as microglia – could slow or stop the proliferation of tau tangles, and potentially delay or prevent Alzheimer’s dementia.
“I think we’ve found a potential link between amyloid and tau that people have been looking for for a long time,” said senior author David Holtzman, MD, the Andrew B. and Gretchen P. Jones Professor and head of the Department of Neurology. “If you could break that link in people who have amyloid deposition but are still cognitively healthy, you might be able to stop disease progression before people develop problems with thinking and memory.”
While the formation of amyloid plaques and tau tangles have been recognized as key steps in the development of Alzheimer’s disease, researchers have struggled to pin down the relationship between the two. By themselves, amyloid plaques do not cause dementia. Many people over age 70 have some amyloid plaques in their brains, including some who are as mentally sharp as ever. But the presence of amyloid plaques seems to lead inexorably to the formation of tau tangles – the true villain of Alzheimer’s – and, until now, it wasn’t clear how amyloid drives tau pathology.
Holtzman and colleagues – including first authors Cheryl Leyns, PhD, a former graduate student in Holtzman’s lab, and Maud Gratuze, PhD, a postdoctoral researcher, as well as co-senior author Jason Ulrich, PhD, an assistant professor of neurology – suspected that microglia could be the link. A rare mutation in a gene called TREM2 leaves people with weak and ineffective microglia, and also increases their risk of developing Alzheimer’s by twofold to fourfold.